Unzipping Nanotubes Graphics
On this page you will find the graphics from the Nature Paper “Longitudinal Unzipping of Carbon Nanotubes to Form Graphene Nanoribbons†(DOI 10.1038/nature07872). Click on the links in the caption for each set of images to download the image.


AFM images (with no caption, with caption) of partially stacked multiple short fragments of nanoribbons that were horizontally cut by tip-ultrasonic treatment of the original oxidation product to facilitate spin-casting onto the mica surface. From the height data (inset), the ribbons are generally single-layered with straight edges. Some stacking is observed even in the oxidized form. The two smaller figures to the right show some other characteristic nanoribbons. Images by: B. Katherine Price and Amanda L. Higginbotham
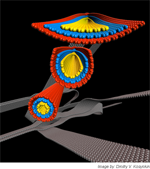
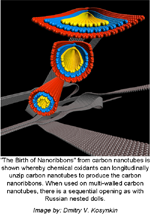


Birth of nanoribbon (black background, no caption; black background, with caption; gradient background, no caption; gradient background, with caption) – “The Birth of Nanoribbons” from carbon nanotubes is shown whereby chemical oxidants can longitudinally unzip carbon nanotubes to produce the carbon nanoribbons. When used on multi-walled carbon nanotubes, there is a sequential opening as with Russian nested dolls. Image by: Dmitry V. Kosynkin

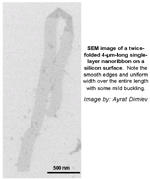
SEM image (without caption, with caption) of a twice-folded 4-μm-long single-layer nanoribbon on a silicon surface. Note the smooth edges and uniform width over the entire length with some mild buckling. Image by: Ayrat Dimiev
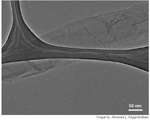
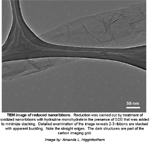
TEM image of reduced nanoribbons (without caption, with caption). Reduction was carried out by treatment of oxidized nanoribbons with hydrazine monohydrate in the presence of SDS that was added to minimize stacking. Detailed examination of the image reveals 2-3 ribbons are stacked with apparent buckling. Note the straight edges. The dark structures are part of the carbon imaging grid. Image by: Amanda L. Higginbotham


TEM images (without caption, with caption) depicting the transformation of multi-walled carbon nanotubes (MWCNTs) into oxidized nanoribbons. The starting material (left) consists of regular MWCNT structures that are 40 – 80 nm in diameter. After treatment with KMnO4 in sulfuric acid at 23 – 70 °C for 2 h, nanoribbons (right) are formed whose surface closely resembles that of graphite oxide but with straight edges. The right side of the ribbon is partially folded onto itself. The dark structures are part of the carbon imaging grid. Images by: Amanda L. Higginbotham
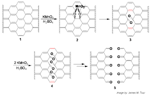

Chemical mechanism of carbon nanotube opening (without caption, with caption (the larger file is much better resolution than the thumbnail above; also ChemDraw 10 or Encapsulated PostScript files). A section of the nanotube sidewall is shown in 1. The initial wall in 1 is subject to permanganate oxidation to afford the manganate ester 2 [(RO)2MnO2- or (RO)2MnO2H if protonated] which can oxidize further to the dione 3 in the dehydrating medium. The severe steric hindrance induced by the juxtaposed ketones in 3 distorts the β,γ-alkenes (red) making them more prone to the next attack by permanganate resulting in 4 and eventually to the fully cut nanotube 5. Image by: James M. Tour


Carbon nanotube unzipping (without caption, with caption). Schematic representation of the gradual unzipping of one wall of a carbon nanotube to form a nanoribbon. Although depicted here as an internal rather than end-origin of the initial attack, the location of initial attack may vary. Image by: Dmitry V. Kosynkin
Additional Images

Six tubes, unzipping from left to right, signed by the artist Dmitry V. Kosynkin (2 MB)
A version of the cover in gradient black, signed by the artist Dmitry V. Kosynkin (3 MB)

The Nature Cover (2 MB), copyright Macmillan Publilshers.
A Power Point file with three figures from the Nature Paper(7 MB)
